Streptomyces Bacteriophages
Streptomyces bacteriophages
Prof Paul A Hoskisson, University of Strathclyde.
Introduction
Bacteriophages (or phages) are amongst the most abundant biological entities on the planet and exhibit enormous diversity[1]. There has been a huge amount of interest in Streptomyces phages over many years as they have been a source of many valuable tools for molecular biology and genetic manipulation. These include the widely used integrating vector systems[2] based on phage integrases, such as pSET152, pMS82 and their derivatives, along with newer integrase systems[3–5], mutagenesis[6] and the older, seldom-used nowadays, phage cloning vectors[7,8]. Streptomyces bacteriophages have also been extensively studied from the perspective of their own biology and diversity[1,9,10], and also in Streptomyces from the perspective of bacteriophage resistance[11,12].
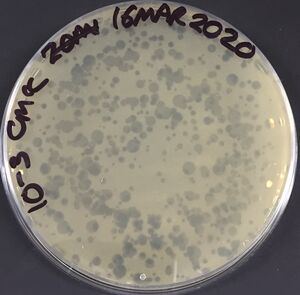
Streptomyces phages are readily isolated from a range of soil and environmental samples on soft agar overnight (see protocol for Isolation of phages from environmental samples), with isolation of novel phage from single phages easily achieved. Success can be increased through enrichment procedures (see protocol Isolation of phages from environmental samples), with the isolation of lytic and temperate phages being relatively straightforward.
Phages can also readily be induced from lysogens (prophages) using the standard methods of UV-induction, mitomycin C-induction and from spontaneous induction events following growth in liquid medium, which provides free phage for plaque formation on suitable indicator strains (not always straightforward as endogenous phage often have narrow host ranges and the original strain is likely immune to a phage it carries).
The model Streptomyces phage (our equivalent of the model coliphage, λ) is the temperate phage ΦC31, with clear plaque (lytic) mutants (cΔ25) available and often used in the laboratory. The R4-like group of phages appear to be the most abundant Streptomyces phages globally[1]. Beyond the Streptomycetes there have been limited investigations of phage within the Actinobacteria - the exception being the extensive work on Mycobacteriophages[10,13,14] and Corynephages[15].
Protocols
Streptomyces Bacteriophage plaque assay
High titre preparation of phage
Isolation of phages from single plaques
Isolation of phages from environmental samples
References
1. Smith, M. C. M. et al. Evolutionary relationships among actinophages and a putative adaptation for growth in Streptomyces spp. J Bacteriol 195, 4924–35 (2013).
2. Baltz, R. H. Streptomyces temperate bacteriophage integration systems for stable genetic engineering of actinomycetes (and other organisms). J Ind Microbiol Biot 39, 661–72 (2011).
3. Bierman, M. et al. Plasmid cloning vectors for the conjugal transfer of DNA from Escherichia coli to Streptomyces spp. Gene 116, 43–49 (1992).
4. Gregory, M., Till, R. & Smith, M. Integration Site for Streptomyces Phage BT1 and Development of Site-Specific Integrating Vectors. Journal of Bacteriology 185, (2003).
5. Fogg, P. C. M., Haley, J. A., Stark, W. M. & Smith, M. C. M. Genome Integration and Excision by a New Streptomyces Bacteriophage, ϕJoe. Appl Environ Microb 83, e02767-16 (2017).
6. Khodakaramian, G. Expression of Cre recombinase during transient phage infection permits efficient marker removal in Streptomyces. Nucleic Acids Research 34, (2006).
7. Foor, F. & Morin, N. Construction of a shuttle vector consisting of the Escherichia coli plasmid pACYC177 inserted into the Streptomyces cattleya phage TG1. Gene 94, 109–113 (1990).
8. Zhou, X., Deng, Z., Hopwood, D. A. & Kieser, T. Characterization of phi HAU3, a broad-host-range temperate streptomyces phage, and development of phasmids. J Bacteriol 176, 2096–2099 (1994).
9. Kieser, T. et al. Practical Streptomyces Genetics. (2000).
10. Hendrix, R. W., Smith, M. C., Burns, R. N., Ford, M. E. & Hatfull, G. F. Evolutionary relationships among diverse bacteriophages and prophages: all the world’s a phage. Proceedings of the National Academy of Sciences of the United States of America 96, 2192–7 (1999).
11. Hoskisson, P. A. & Smith, M. C. Hypervariation and phase variation in the bacteriophage ‘resistome’. Current Opinion in Microbiology 10, (2007).
12. Hoskisson, P. A., Sumby, P. & Smith, M. C. M. The phage growth limitation system in Streptomyces coelicolor A(3)2 is a toxin/antitoxin system, comprising enzymes with DNA methyltransferase, protein kinase and ATPase activity. Virology (2015) doi:10.1016/j.virol.2014.12.036.
13. Kessel, J. C. van & Hatfull, G. F. Efficient point mutagenesis in mycobacteria using single-stranded DNA recombineering: characterization of antimycobacterial drug targets. Molecular Microbiology 67, (2008).
14. Hatfull, G. F. Molecular Genetics of Mycobacteria. 81–119 (2014) doi:10.1128/9781555818845.ch5.
15. Sangal, V. & Hoskisson, P. A. Corynephages: Infections of the Infectors. In: Corynebacterium diphtheriae and Related Toxigenic Species. pp67–81; Springer; Edited Burkovski, A. (2013). doi:10.1007/978-94-007-7624-1_4.